The FDA recall of a medical device last week has caused a bit of a media storm as the general public scrambles to find out more. The fact that a medical device meant to help sustain life is insecure and could be hacked to kill a patient is alarming to all of us. More worrying is that the medical device subject to the recall, a cardiac rhythm management product, or “pacemaker” to the rest of us, is probably not an anomaly. Many other medical devices more than likely also lack adequate security.
To understand the risks, we first need to understand the problem. To be honest, this could require an extensive series of blog posts over weeks to fully examine and explain this properly, but here’s the 50,000-foot version.
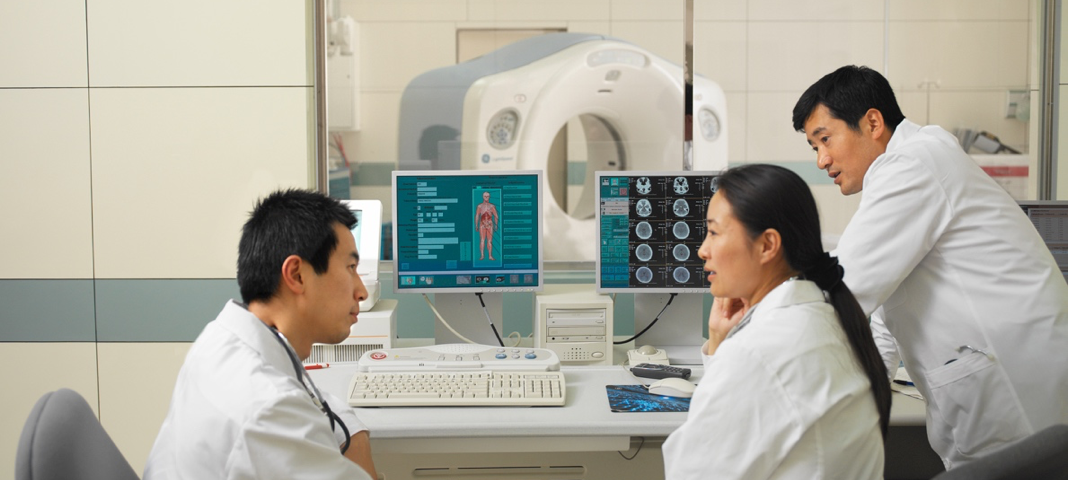
Different types of medical devices and the risks they pose
First, there are the implantable medical devices (IMDs) like the medical pacemaker at the center of this story. This group of medical devices includes the implanted insulin pump that security researcher Barnaby Jack demonstrated live on stage at the Miami Hacker Halted Conference in 2011, hacking the device to deliver a lethal drug dose. It also includes a pacemaker that was hacked, again by Jack, at the Melbourne BreakPoint Security Conference in 2012 to deliver a lethal 830 volt electric shock to a patient.
Second are the much wider range of network-attached medical devices used in healthcare delivery. These include:
- Diagnostic imaging systems: ultrasound, MRI, PET, CT scanners, and X ray machines
- Treatment equipment: infusion pumps, medical lasers, and surgical machinery
- Life support: ventilators, anesthetic and dialysis machines
- Medical monitors for oxygen saturation, blood pressure, ECG and EEG, and many, many more.
<break>
The greatest data-security risks for medical devices
The network-attached group far outnumbers the IMD group, but both have one thing in common—a very long life span! No one wants a pacemaker that needs to be replaced every year or two, and hospitals simply can’t afford to rip and replace their multimillion-dollar investment in x-ray machines and PET and CT scanners if they still work perfectly. Many current medical devices are 15 or 20 years old already, placed into service when the rest of us were deploying Windows 95 and dial-up modems.
The greatest risk to medical devices, however, is that they lack even the basic security protections that a $200 home PC has—things like antivirus software and a host firewall. The danger is that when a malware worm gets into a hospital and spreads its way laterally across the network to reach highly vulnerable medical devices, it either quickly infects them (many of the newer models run a form of Windows XP), or the malware multicast traffic storm causes the medical device to crash or just stop working. It’s not that someone hacked and changed a parameter—although that is a distinct possibility—but it’s more likely that its battery becomes quickly drained and powers off, or the system blue screens and ceases to provide life-sustaining care.
It’s going to take years to patch or replace the arsenal of insecure medical devices and billions of dollars that healthcare providers simply don’t have. So, we need to look at alternatives to secure them for the rest of their life-spans.
How to reduce risk and protect devices
By far the most effective approach is microsegmentation, where medical devices are locked down and secured by the network they are attached to. (Attempting to manage 350,000 individual medical devices in a hospital is impossible.)
Modern network infrastructure supports security technologies like Cisco TrustSec©, where each network port acts as a virtual firewall. Using security group tags, network traffic is controlled so that only specifically authorized users—biomedical equipment technicians (or BMETs, as they are known)—have access to reprogram devices, and these systems are only able to communicate with designated internal IP addresses using predetermined ports and protocols. The network will drop everything else, like malware traffic and any connection attempts from unauthorized users. Many of the more advanced healthcare providers have already adopted such an approach, and by employing compensating security controls like TrustSec have been able to secure their networked medical devices from attack.
Learn more about Cisco’s approach towards medical device security
Find out if TrustSec and Microsegmentation are right for you
For more information on cybersecurity solutions, get the details on Cisco’s Digital Network Architecture for Healthcare and IoT Threat Defense for network-connected devices.






Once medical devices are authenticated, continuous monitoring and visibility for all network access, violations, and vulnerabilities is also critical. Use of a Managed Detection and Response(MDR)solution, such as Cisco Active Threat Analytics (ATA) can detect threats within an average of 17 hours vs. the industry average of 100 days through netFlow, metadata and full packet capture (PCAP) analysis. Leveraging Hadoop, statistical as well as behavioral analysis, Cisco ATA can detect medical device anomalous behavior if the devices are compromised after they authenticate to the hospital network.
Thanks Stew and yes, a layered approach to security is always going to be better. The more you can see the better you can secure. NetFlow analysis, combined with ISE / TrustSec object and identity management controls will help immediately identity medical devices, and behavior analysis and anomaly detection will identify any strange behavior that could be an early indicator of compromise.
When medicinal gadgets are confirmed, consistent checking and perceive ability for all system access, infringement, and vulnerabilities are additionally basic by the Use of a Managed Detection and Response arrangement. The security framework can distinguish medicinal gadget atypical conduct if the gadgets are traded off after they verify to the doctor's facility organize.
MDR and security segmentation play a critical part in helping to secure medical devices. Its by far the single biggest headache for healthcare CISOs and CIOs currently. You need visibility of a potential indicator of co promise and at the same time some preventative controls to restrict who and what has access to medical devices.